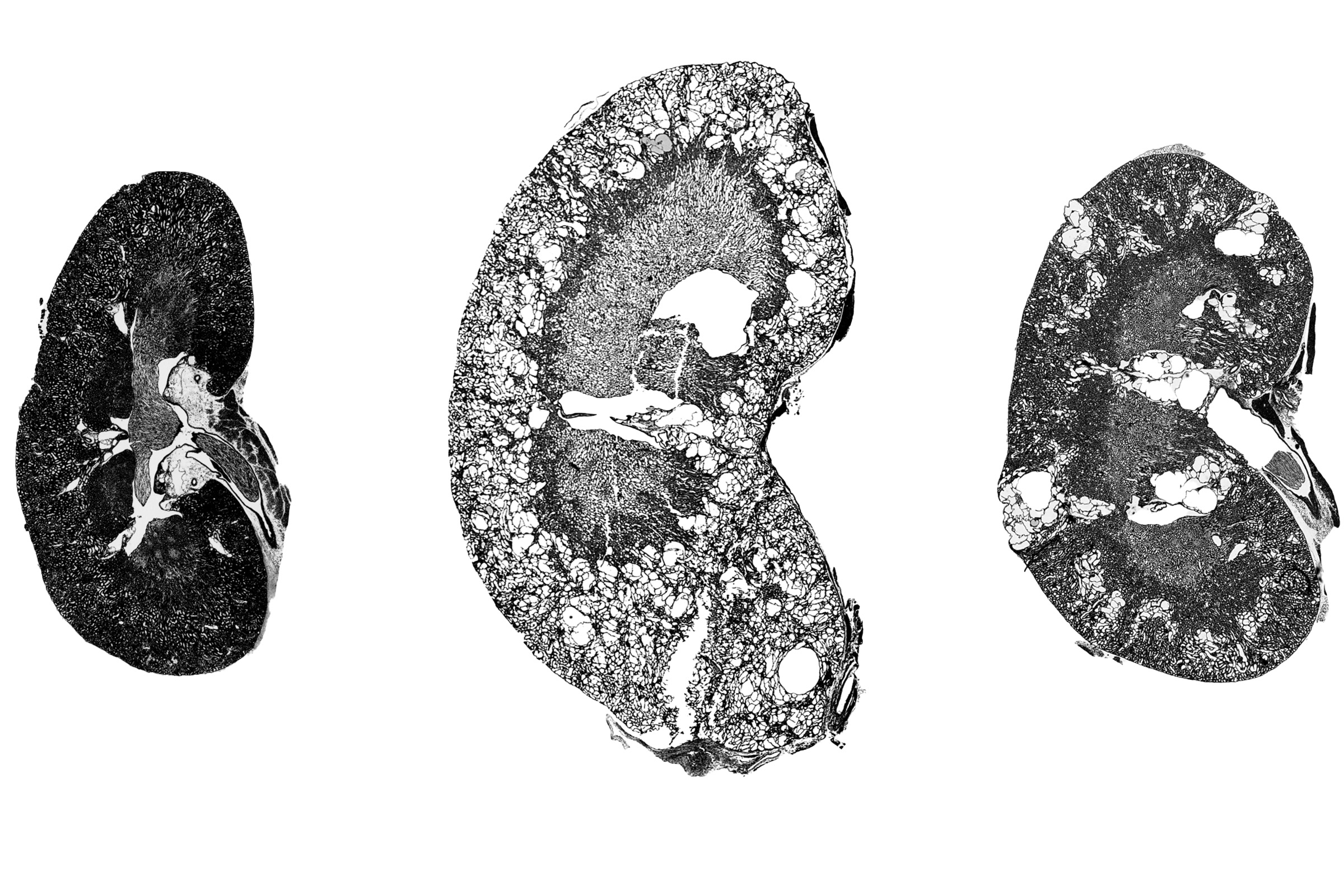
A new drug candidate can shrink kidney cysts
A compound originally developed to treat cancer could be repurposed to treat polycystic kidney disease, an inherited condition that can lead to kidney failure.
Autosomal dominant polycystic kidney disease (ADPKD), the most common form of polycystic kidney disease, can lead to kidney enlargement and eventual loss of function. The disease affects more than 12 million people worldwide, and many patients end up needing dialysis or a kidney transplant by the time they reach their 60s.
Researchers at MIT and Yale University School of Medicine have now found that a compound originally developed as a potential cancer treatment holds promise for treating ADPKD. The drug works by exploiting kidney cyst cells’ vulnerability to oxidative stress — a state of imbalance between damaging free radicals and beneficial antioxidants.
In a study employing two mouse models of the disease, the researchers found that the drug dramatically shrank kidney cysts without harming healthy kidney cells.
“We really believe this has potential to impact the field and provide a different treatment paradigm for this important disease,” says Bogdan Fedeles, a research scientist and program manager in MIT’s Center for Environmental Health Sciences and the lead author of the study, which appears this week in the Proceedings of the National Academy of Sciences.
John Essigmann, the William R. and Betsy P. Leitch Professor of Biological Engineering and Chemistry at MIT; Sorin Fedeles, executive director of the Polycystic Kidney Disease Outcomes Consortium and assistant professor (adjunct) at Yale University School of Medicine; and Stefan Somlo, the C.N.H. Long Professor of Medicine and Genetics and chief of nephrology at Yale University School of Medicine, are the senior authors of the paper.
Cells under stress
ADPKD typically progresses slowly. Often diagnosed when patients are in their 30s, it usually doesn’t cause serious impairment of kidney function until patients reach their 60s. The only drug that is FDA-approved to treat the disease, tolvaptan, slows growth of the cysts but has side effects that include frequent urination and possible liver damage.
Essigmann’s lab did not originally set out to study PKD; the new study grew out of work on potential new drugs for cancer. Nearly 25 years ago, MIT research scientist Robert Croy, also an author of the new PNAS study, designed compounds that contain a DNA-damaging agent known as an aniline mustard, which can induce cell death in cancer cells.
In the mid 2000s, Fedeles, then a grad student in Essigmann’s lab, along with Essigmann and Croy, discovered that in addition to damaging DNA, these compounds also induce oxidative stress by interfering with mitochondria — the organelles that generate energy for cells.
Tumor cells are already under oxidative stress because of their abnormal metabolism. When they are treated with these compounds, known as 11beta compounds, the additional disruption helps to kill the cells. In a study published in 2011, Fedeles reported that treatment with 11beta compounds significantly suppressed the growth of prostate tumors implanted in mice.
A conversation with his brother, Sorin Fedeles, who studies polycystic kidney disease, led the pair to theorize that these compounds might also be good candidates for treating kidney cysts. At the time, research in ADPKD was beginning to suggest that kidney cyst cells also experience oxidative stress, due to an abnormal metabolism that resembles that of cancer cells.
“We were talking about a mechanism of what would be a good drug for polycystic kidney disease, and we had this intuition that the compounds that I was working with might actually have an impact in ADPKD,” Bogdan Fedeles says.
The 11beta compounds work by disrupting the mitochondria’s ability to generate ATP (the molecules that cells use to store energy), as well as a cofactor known as NADPH, which can act as an antioxidant to help cells neutralize damaging free radicals. Tumor cells and kidney cyst cells tend to produce increased levels of free radicals because of the oxidative stress they’re under. When these cells are treated with 11beta compounds, the extra oxidative stress, including the further depletion of NADPH, pushes the cells over the edge.
“A little bit of oxidative stress is OK, but the cystic cells have a low threshold for tolerating it. Whereas normal cells survive treatment, the cystic cells will die because they exceed the threshold,” Essigmann says.
Shrinking cysts
Using two different mouse models of ADPKD, the researchers showed that 11beta-dichloro could significantly reduce the size of kidney cysts and improve kidney function.
The researchers also synthesized a “defanged” version of the compound called 11beta-dipropyl, which does not include any direct DNA-damaging ability and could potentially be safer for use in humans. They tested this compound in the early-onset model of PKD and found that it was as effective as 11beta-dichloro.
In all of the experiments, healthy kidney cells did not appear to be affected by the treatment. That’s because healthy cells are able to withstand a small increase in oxidative stress, unlike the diseased cells, which are highly susceptible to any new disturbances, the researchers say. In addition to restoring kidney function, the treatment also ameliorated other clinical features of ADPKD; biomarkers for tissue inflammation and fibrosis were decreased in the treated mice compared to the control animals.
The results also suggest that in patients, treatment with 11beta compounds once every few months, or even once a year, could significantly delay disease progression, and thus avoid the need for continuous, burdensome antiproliferative therapies such as tolvaptan.
“Based on what we know about the cyst growth paradigm, you could in theory treat patients in a pulsatile manner — once a year, or perhaps even less often — and have a meaningful impact on total kidney volume and kidney function,” Sorin Fedeles says.
The researchers now hope to run further tests on 11beta-dipropyl, as well as develop ways to produce it on a larger scale. They also plan to explore related compounds that could be good drug candidates for PKD.
Other MIT authors who contributed to this work include Research Scientist Nina Gubina, former postdoc Sakunchai Khumsubdee, former postdoc Denise Andrade, and former undergraduates Sally S. Liu ’20 and co-op student Jake Campolo. The research was funded by the PKD Foundation, the U.S. Department of Defense, the National Institutes of Health, and the National Institute of Environmental Health Sciences through the Center for Environmental Health Sciences at MIT.