Wendlandt paper published in Journal of the American Chemical Society
The paper, A Change from Kinetic to Thermodynamic Control Enables trans-Selective Stereochemical Editing of Vicinal Diol, was published on December 20, 2021
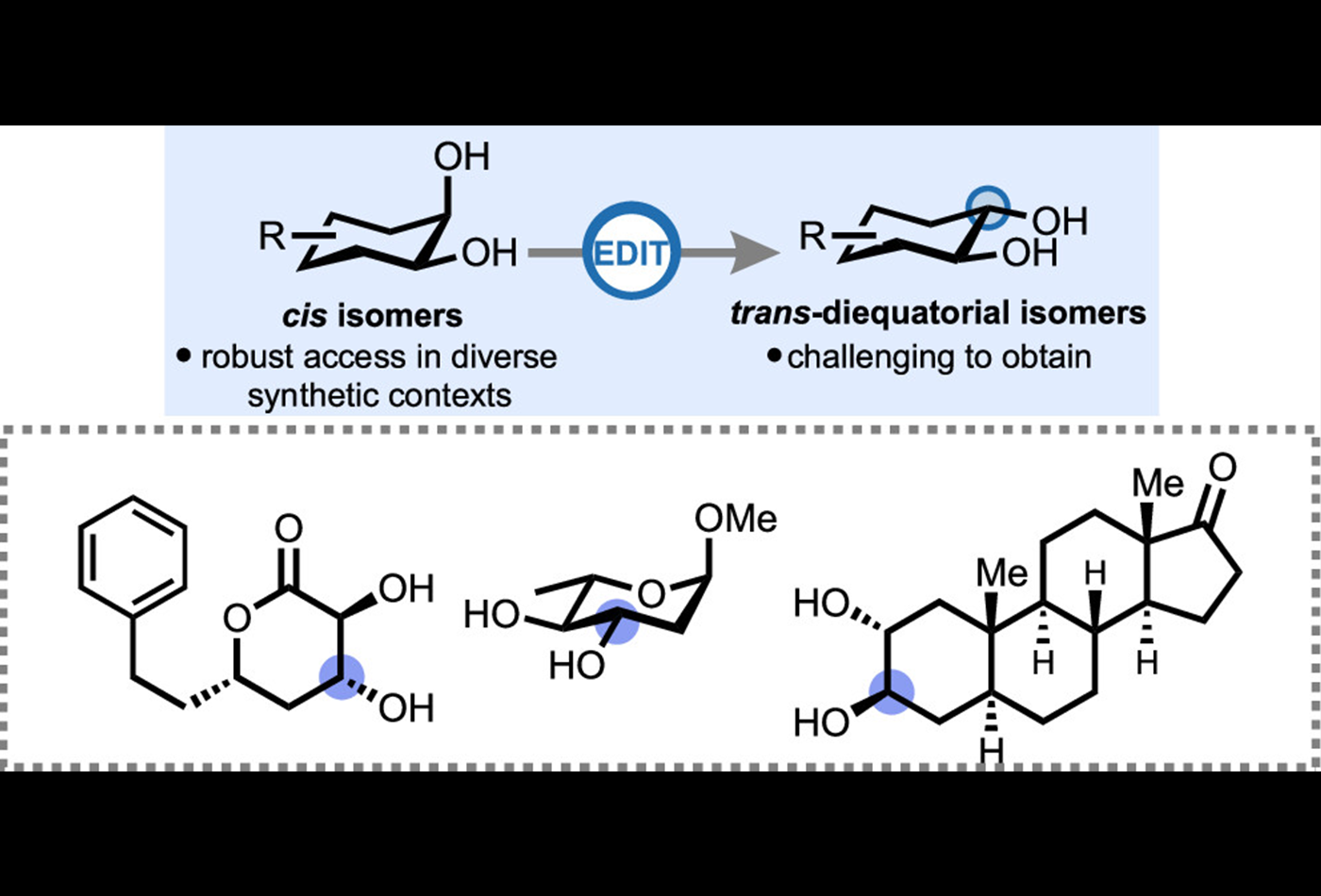
A paper authored by Yu-An Zhang, Xin Gu, and Professor Alison E. Wendlandt was published in Journal of the American Chemical Society on December 20, 2021.
A Change from Kinetic to Thermodynamic Control Enables trans-Selective Stereochemical Editing of Vicinal Diols
Yu-An Zhang, Xin Gu, and Alison E. Wendlandt
J. Am. Chem. Soc.
Published 20 December 2021
DOI: https://doi.org/10.1021/jacs.1c11902
Abstract
Here, we report the selective, catalytic isomerization of cis-1,2-diols to trans-diequatorial-1,2-diols. The method employs triphenylsilanethiol (Ph3SiSH) as a catalyst and proceeds under mild conditions in the presence of a photoredox catalyst and under blue light irradiation. The method is highly chemoselective, broadly functional group tolerant and provides concise access to trans-diol products which are not readily obtained using other methods. Mechanistic studies reveal that isomerization proceeds through a reversible hydrogen atom transfer pathway mediated by the silanethiol catalyst.
Research in the Wendlandt Group focuses on the development of selective, catalytic reactions using the tools of organic/organometallic synthesis and physical organic chemistry.