Study reveals the dynamics of human milk production
A new analysis shows how milk-producing cells change over time in nursing mothers.
For the first time, MIT researchers have performed a large-scale, high-resolution study of the cells in breast milk, allowing them to track how these cells change over time in nursing mothers.
By analyzing human breast milk produced between three days and nearly two years after childbirth, the researchers were able to identify a variety of changes in gene expression in mammary gland cells. Some of these changes were linked to factors such as hormone levels, illness of the mother or baby, the mother starting birth control, and the baby starting daycare.
“We were able to take this really long view of lactation that other studies haven’t really done, and we showed that milk does change over the entire course of lactation, even after years of milk production,” says Brittany Goods, a former MIT postdoc who is now an assistant professor of engineering at Dartmouth College, and one of the senior authors of the study.
The researchers hope that their findings will lay the groundwork for more in-depth studies of how breast milk changes over time. Such studies could eventually yield new ways to boost mothers’ milk production or to improve the composition of infant formula.
Bonnie Berger, the Simons Professor of Mathematics at MIT and head of the Computation and Biology group at the Computer Science and Artificial Intelligence Laboratory (CSAIL), is a senior author of the study, as is Alex Shalek, an associate professor of chemistry at MIT and a member of the Institute for Medical Engineering and Science (IMES); the Koch Institute for Integrative Cancer Research; the Ragon Institute of MGH, MIT and Harvard; and the Broad Institute of Harvard and MIT.
MIT graduate student Sarah Nyquist is the lead author of the paper, which appears this week in the Proceedings of the National Academy of Sciences.
Cellular changes
Human mammary glands can produce more than a liter of milk in a day, for months or years after childbirth. Studying how mammary gland cells accomplish this feat has been difficult in humans because the tissue itself can’t be biopsied or otherwise accessed during lactation. However, recent studies have shown that breast milk contains many cells from the mammary gland, offering a noninvasive way to study these cells.
For this study, the MIT team collected breast milk samples from 15 nursing mothers. Each donor provided samples at multiple time points, ranging from three to 632 days after giving birth. The researchers also collected information about health and lifestyle changes that occurred throughout the lactation period.
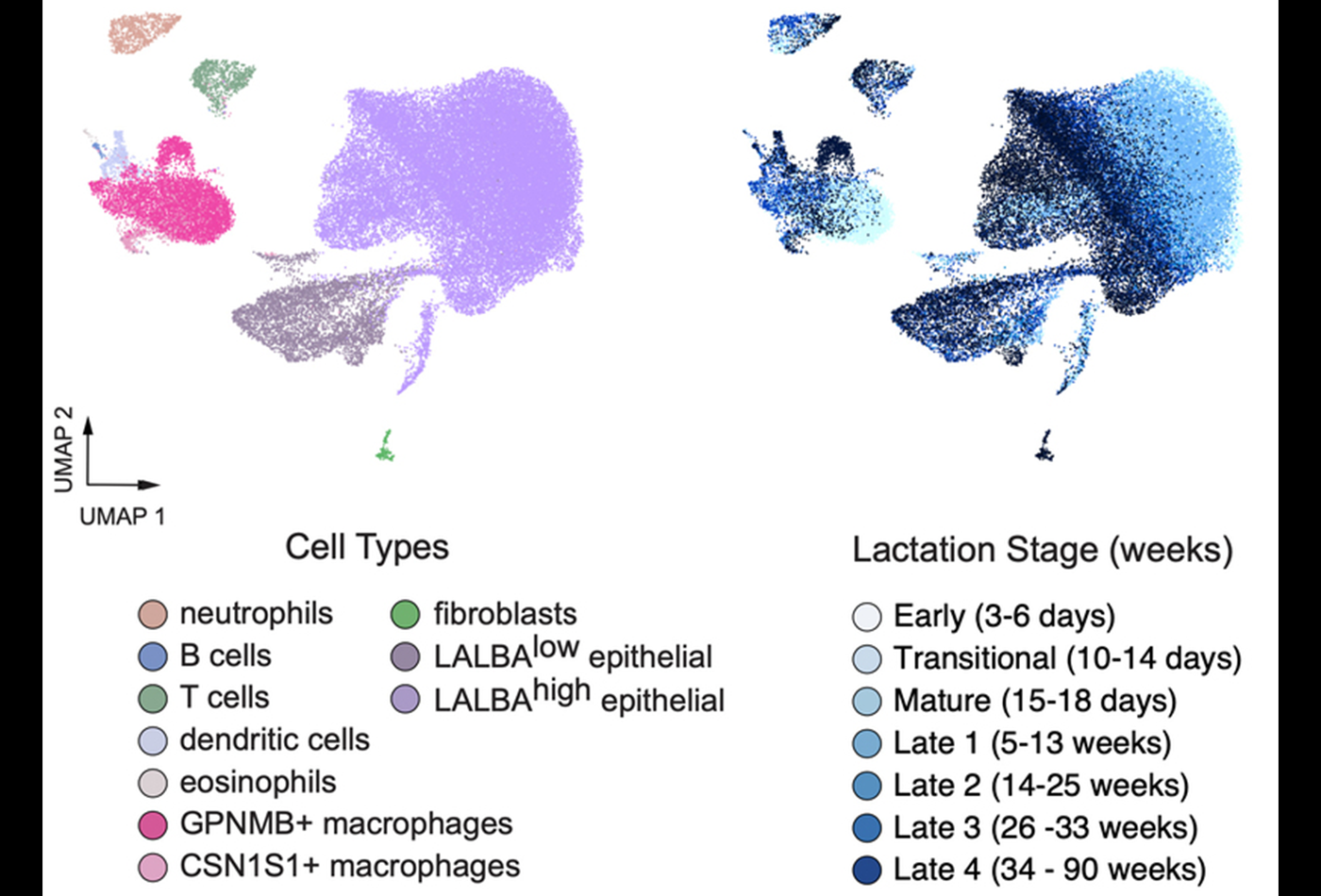
The researchers isolated more than 48,000 cells from 50 samples and analyzed them using single-cell RNA-sequencing, a technology that can determine which genes are being expressed in a cell at a given moment in time. This analysis revealed 10 types of cells — a population of fibroblast cells, two types of epithelial cells, and seven types of immune cells.
More than half of the immune cells that they found were macrophages. These cells appear to express genes that help make the mammary gland more tolerant of the milk proteins that they are producing, so they don’t trigger an immune response. The researchers also found populations of B cells, T cells, and other immune cells, but their numbers were too small to do any in-depth studies of their functions.
By far the most abundant cells that they found were lactocytes, which are a type of epithelial cell. These cells expressed many genes for proteins that are found in breast milk, such as lactalbumin, as well as transporters needed to secrete milk proteins, micronutrients, fat, and other breast milk components.
Among the lactocytes, the researchers identified one cluster of cells that appears to be the primary producer of milk, and another that plays more of a structural role in the mammary gland. Each of these cell types could be divided into further subtypes, which the researchers hypothesize may be specialized for particular roles.
As time went on, the researchers found that the proportion of lactocytes involved in milk production went down, while the proportion involved in structural support went up. At the same time, genes involved in responding to the hormone prolactin became more active in the milk-producing lactocytes but dropped off in structural lactocytes. The researchers theorize that these changes may be related to the changing nutritional needs of infants as they grow.
“This study, along with some other studies that are out there, paves the way for mapping out and better understanding some of the pathways that these cells use to accomplish the tremendous amount of work that they do,” Goods says.
Milk composition
The researchers also found links between the composition of cells in breast milk and events such as babies starting to go to daycare, starting formula, or the mother starting to use hormonal birth control.
“There are clearly changes in the composition of breast milk that are related to these lifestyle and health changes, such as infant illness or maternal hormonal birth control,” Nyquist says. “These changes in lactation don’t necessarily have a positive or negative impact on anyone’s health, but they do occur and they may lead us to insights into how mammary epithelial cells are producing milk and the types of components that they may be producing.”
The researchers now hope to do larger studies that could help them find stronger links between environmental factors and milk composition, and also discover more about how milk naturally changes over time. This could eventually help scientists devise better infant formulas or create formulas adapted to different stages of infancy. The researchers also hope to find ways to help nursing mothers boost their milk production or slow it down when babies are being weaned.
Other follow-up studies may explore how pumping affects milk composition and breast health, or how to prevent conditions such as mastitis.
“By building this really high-resolution understanding of lactational diversity over time, it gives us a way to not only understand lactation, but it also gives us a set of data and tools to be able to engineer better solutions to improve the quality of life of mothers, specifically when they’re nursing,” Goods says.
The research was funded by the Koch Institute Support (core) Grant from the National Cancer Institute, the National Institutes of Health, a National Research Service Award postdoctoral fellowship, the Beckman Young Investigator Program, a Sloan Fellowship in Chemistry, the Charles E. Reed Faculty Initiative, a National Science Foundation Graduate Research Fellowship, an MIT-GSK Gertrude B. Elion Postdoctoral Fellowship, the Columbia University Office of the Provost, the Weizmann Institute of Science National Postdoctoral Award Program for Advancing Women in Science, the International Society for Research in Human Milk and Lactation Trainee Bridge Fund, and the Human Frontier Science Program.