Single-molecule tracker illuminates workings of cancer-related proteins
Researchers use custom-built microscopy and nanotechnology to tag and follow the activity of individual proteins in real time.
Highlights
- Researchers developed a single-molecule imaging technology that can track individual proteins in living cells over long periods of time.
- In the new study, they used the method to follow EGFR family receptors, which are implicated in several cancers, in human cells. They provide an unprecedented view of how the receptors pair and unpair over time, revealing unexpected receptor dynamics.
- The method could be used to investigate other kinds of molecules and potentially drugs to better understand how they impact signaling within the cell.
Using a powerful single-molecule imaging method they developed, a Broad Institute research team has unveiled a dynamic view of how some cancer-related proteins interact in living cells. The technique relies on highly stable nanoparticle probes that brightly illuminate individual molecules for long periods of time. The researchers used their method to observe, for the first time, individual receptors as they move around the cell membrane, attaching to and then letting go of other receptors to alter signaling within the cell.
Described in Cell, the work demonstrates the method’s potential for investigating other receptors and molecules and for improved drug screening to better understand the effects of therapeutics on living cells.
“With our photostable probes, we can map out the entire lifespan of these molecules in their native environment and see things that have never been observable before,” said study leader Sam Peng, a Broad core institute member and assistant professor of chemistry at MIT.
Molecular movies
Peng’s method solves a problem with existing contrast agents used in single-molecule tracking such as dyes. Under the laser light that’s used to excite these dyes, they burn out after a few seconds in a phenomenon known as photobleaching, which means that scientists could only use them to take a few snapshots of cell receptors and not follow them over the entirety of the signaling process.
For a longer and richer view, Peng’s lab developed long-lasting probes, known as upconverting nanoparticles, which emit signals that remain stable under laser excitation. The nanoparticles contain rare-earth ions that continue to luminescence for minutes, hours, and potentially years. In addition, by altering the type and doses of the ions, scientists can engineer probes emitting in many different colors, enabling tracking of many targets in a single experiment.
In the current study, the researchers aimed to uncover new biology by focusing on the EGFR family of cell receptors, which have been linked to several kinds of cancer. They collaborated with EGFR experts Matthew Meyerson and Heidi Greulich of the Broad’s Cancer Program. They knew that EGFR receptors need to pair up, or “dimerize,” in order to initiate signaling within the cell, but they wanted to learn more about the dynamics of these pairings — what the receptors partner with, how long they stay together, and how they find new partners.
For a better and more sustained look at the receptors, the research team customized their upconverting nanoparticles to tag EGFR and related receptors HER2 and HER3, which are linked to cancer, and used them to track the molecules in living human cells.
A new view of protein pairings
In this study, Peng and his team observed that, when activated with a stimulating molecule, EGFR receptors can pair up and stay dimerized for several minutes, something not observable using traditional dyes. Excessive and prolonged dimerization can lead to too much cell growth and cancer.
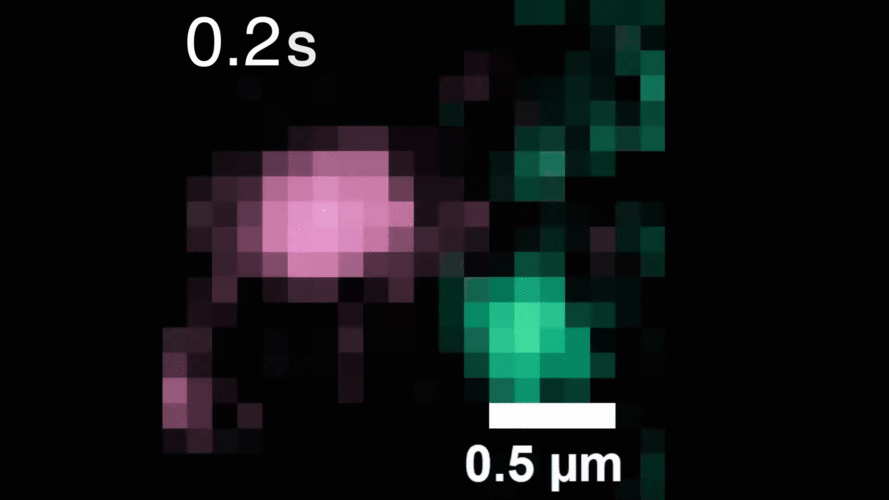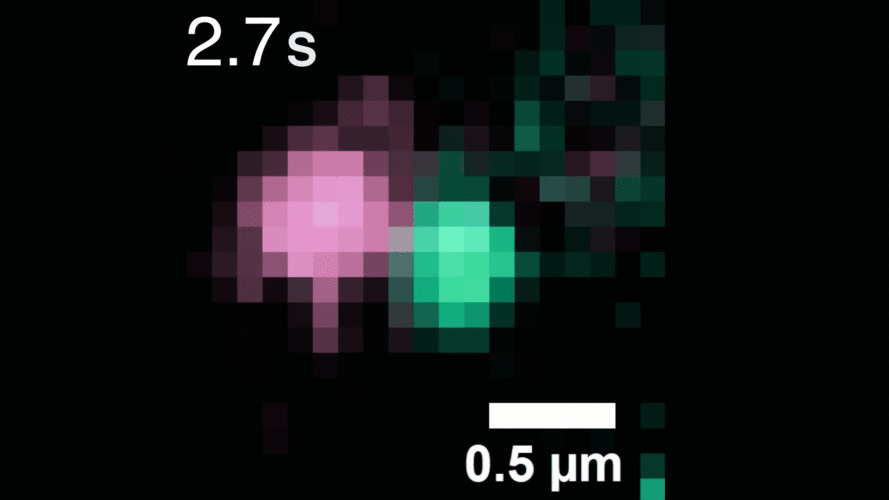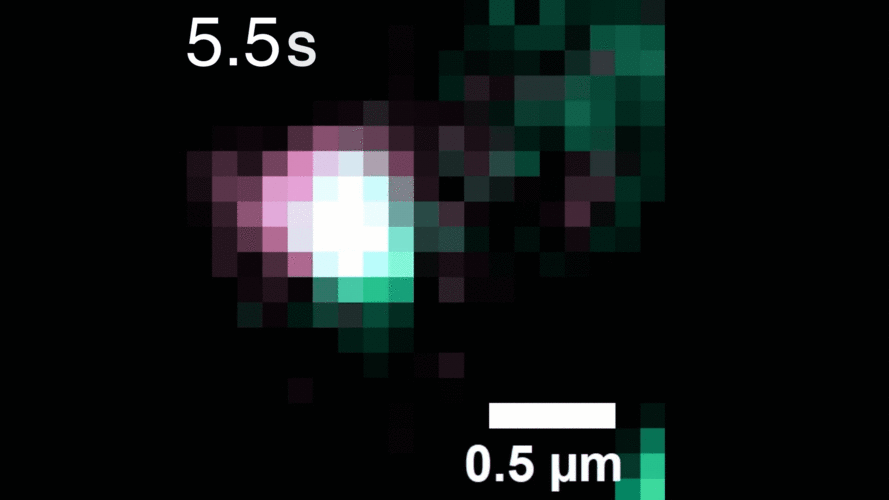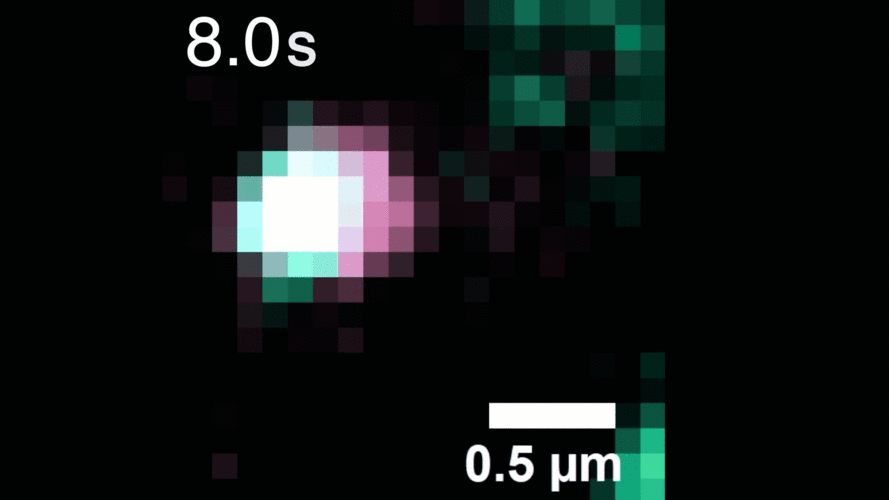
When the EGFR molecules carried cancer-related mutations, the dimers became more stable, with the more stabilizing mutations linked to more potent cancers in people. In addition, the mutated receptors could form stable dimers even without an external stimulus prompting them to dimerize. The finding helps explain how EGFR mutations can lead to uncontrolled cell growth and cancer and could inform efforts to target this process therapeutically.
The team discovered several other new and surprising details about how HER2 and HER3 form stable pairings with themselves, which helps illuminate the role of these molecules in related cancers.
When the research team tagged all three receptor types in one experiment, they observed a vibrant scene with receptors navigating the cell surface, finding partners, unpairing, and then finding new partners, over and over again.
Beyond shedding light on EGFR biology, the scientists hope that collaborators in other fields will apply their method to ask new scientific questions about other proteins of interest. “We think this technique could be transformative for studying molecular biology because it enables dynamic biological processes to be observed with high spatiotemporal resolution over unprecedented timescales,” said Peng.
They are also planning to explore the method’s use in studying the mechanism of drug action, to reveal how potential therapeutics alter individual molecules over time. In addition, they will continue to improve their methods, such as making the probes smaller, brighter, and able to emit more colors.





