Pentelute paper published in Communications Chemistry
The paper, Rapid de novo Discovery of Peptidomimetic Affinity Reagents for Human Angiotensin Converting Enzyme 2, was published on January 19, 2022.
A paper authored by Genwei Zhang, Joseph S. Brown, Anthony J. Quartaroro, Chengxi Li, Xuyu Tan, Stephanie Hanna, Sarah Antilla, Amanda E. Cowfer, Andrei Loas, and Professor Bradley L. Pentelute was published in Communications Chemistry on January 19, 2022.
Rapid de novo discovery of peptidomimetic affinity reagents for human angiotensin converting enzyme 2
Genwei Zhang, Joseph S. Brown, Anthony J. Quartaroro, Chengxi Li, Xuyu Tan, Stephanie Hanna, Sarah Antilla, Amanda E. Cowfer, Andrei Loas, and Professor Bradley L. Pentelute
Communications Chemistry 5
Published 19 January 2022
DOI: https://doi.org/10.1038/s42004-022-00625-3
Abstract
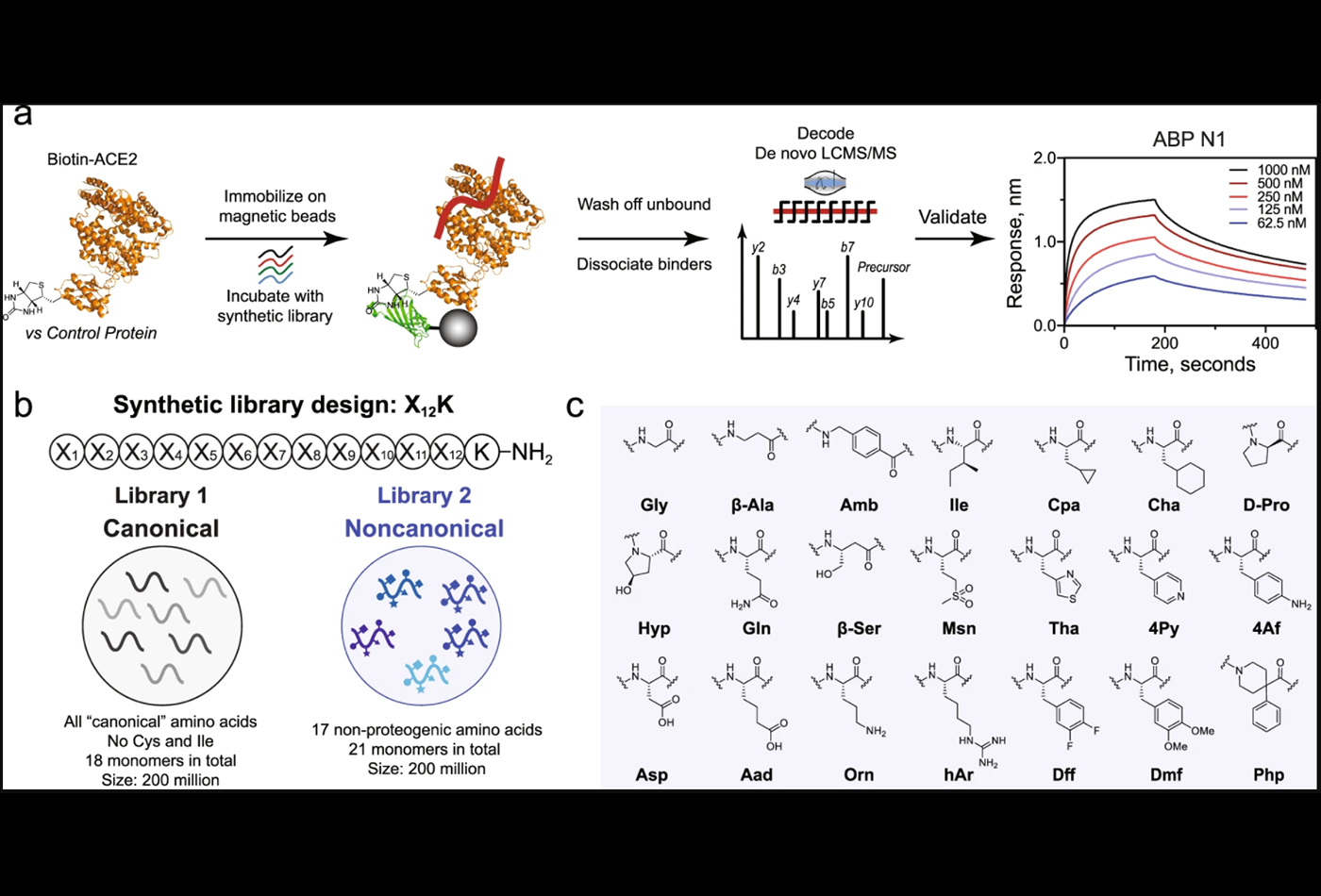
Rapid discovery and development of serum-stable, selective, and high affinity peptide-based binders to protein targets are challenging. Angiotensin converting enzyme 2 (ACE2) has recently been identified as a cardiovascular disease biomarker and the primary receptor utilized by the severe acute respiratory syndrome coronavirus 2. In this study, we report the discovery of high affinity peptidomimetic binders to ACE2 via affinity selection-mass spectrometry (AS-MS). Multiple high affinity ACE2-binding peptides (ABP) were identified by selection from canonical and noncanonical peptidomimetic libraries containing 200 million members (dissociation constant, KD = 19–123 nM). The most potent noncanonical ACE2 peptide binder, ABP N1 (KD = 19 nM), showed enhanced serum stability in comparison with the most potent canonical binder, ABP C7 (KD = 26 nM). Picomolar to low nanomolar ACE2 concentrations in human serum were detected selectively using ABP N1 in an enzyme-linked immunosorbent assay. The discovery of serum-stable noncanonical peptidomimetics like ABP N1 from a single-pass selection demonstrates the utility of advanced AS-MS for accelerated development of affinity reagents to protein targets.
Read the full text on Communications Chemistry
The Pentelute Lab aims to invent new chemistry for the efficient and selective modification of proteins, to ‘hijack’ these biological machines for efficient drug delivery into cells and to create new machines to rapidly and efficiently manufacture peptides and proteins.