Fine-tuning new materials for energy devices
First look at atomic structure reveals path forward
An MIT chemist, Professor Mircea Dincă, and his international collaborators have announced new insights into a remarkable class of highly porous, crystalline materials called metal-organic frameworks, or MOFs. In rare cases, MOFs can be made electrically conductive, producing materials that could enable significant advances in batteries, fuel cells, catalysts, and more. But it hasn’t been clear how to design such MOFs with desired properties or functions. Guided by fundamental studies, the MIT-led researchers were able to grow MOFs as single crystals large enough that a suite of imaging tools could—for the first time—determine their atom-by-atom structure. The researchers could then establish the link between structural details and properties such as electrical conductivity. Based on that information, device designers and others can now select starting materials and synthesis conditions that will produce MOFs optimized for important products and industrial processes.
For more than two decades, much research has focused on a family of materials known as metal-organic frameworks, or MOFs. These special, sponge-like structures are highly porous, have the highest internal surface area of any known material, and generally act as electrical insulators. Moreover, their properties and functions can be altered by changing their composition, their structure, and the conditions under which they are made.
Mircea Dincă, the W.M. Keck Professor of Energy in the Department of Chemistry, has long been intrigued by MOFs and the possibility of designing versions for specific practical uses. In 2015, he and his MIT team developed a way to make MOFs that don’t decompose when exposed to moisture and are therefore suited for use in heat pumps, gas separations and storage, and other industrially important processes. Then, in 2017, they demonstrated MOFs—usually considered electrical insulators—that had electrical conductivity high enough that they could be used in practical devices such as supercapacitors for storing large amounts of energy.
The unusual combination of high porosity and excellent conductivity offered by electrically conductive MOFs has opened up potential applications ranging from batteries, supercapacitors, and fuel cells to electrocatalysts and chemical sensors. However, the development of practical devices has been slowed by the difficulty of readily determining the precise crystalline structure of a given MOF.
“In order to be able to make better materials, you first have to understand what their structure is,” says Dincă. Only with a clear understanding of the atom-by-atom structure of a material can one figure out what aspects of that structure determine various properties. “It’s a fundamental requirement for making better materials.”
So far, there hasn’t been an easy way to get a clear, atomic-level image of the type of MOF structures that show high electrical conductivity. The reason: Getting an image with sufficient detail typically requires growing single crystals. “But single crystals of these particular MOF materials haven’t been large enough to examine using the most common technique, X-ray diffraction imaging,” explains Dincă. “Imaging them with the necessary atomic precision has required using electron diffraction, and not many labs have that capability.”
Back when they first measured high electrical conductivity in their MOFs, Dincă and his team set out to grow MOF crystals large enough to use X-ray diffraction to get the atomic-level information they needed. But that, says Dincă, turned out to be very difficult.
The growth habits of MOFs
The challenge in growing large MOF crystals stems from how the crystals like to grow. A single building block of a MOF is a core made of metal ions (electrically charged particles) surrounded by chemically bound arms, or “ligands,” made of organic (carbon-containing) molecules that radiate outward. Growth occurs when those individual building blocks link together. They can connect side by side to form flat sheets similar to two-dimensional honeycomb sheets of graphene. And they can grow upward, with the flat sheets piling on top of one another. The result then is a regular array of metal ions connected by ligands, which creates a cage-like structure with open pores. For the final result to be a single crystal, all the linkages must occur without any mistakes. A single crystal is, by definition, nearly defect-free.
The problem has been that MOFs grow far more readily in the vertical direction than in the horizontal plane. As a result, single crystals tend to be tall and skinny—and not large enough to analyze using common X-ray diffraction techniques. The usual strategy for controlling how crystals grow involves systematically changing reactant concentrations or reaction temperature and time. But those approaches haven’t altered the directions in which MOFs grow and have yielded only slightly larger crystals.
Dincă and his collaborators realized that they needed to find a way to control the fundamental mechanisms that lead to different growth rates in different directions. For growth to happen, linkages need to break and re-form; those that break more easily are said to be more reversible. The chemical bonds between the metal ions and the ligands in the horizontal sheets are far stronger than the connections that link the stacked up sheets together. Because they’re more reversible, the connections controlling vertical growth break and form at a faster rate than the bonds controlling horizontal growth do. As a result, upward growth exceeds horizontal spreading.
In their earlier work on conducting MOFs, Dincă and his team had focused largely on a single particularly promising ligand molecule. But now they needed to find one with characteristics that would elicit a different growth behavior. For example, they could use a molecule with a different distribution of electrons, that is, exactly where the positive and negative charges occur in its physical structure. “Because plus will attract minus and minus will attract plus, how that distribution occurs within a molecule influences how copies of that molecule will arrange themselves with respect to each other—and that ultimately dictates how the molecules stack up and connect with each other,” explains Dincă.
They could also choose a ligand molecule with lower electron density in certain regions, thereby making it more acidic. If the molecule binding with the metal is more acidic, it will more easily lose a proton (a charged hydrogen atom), leaving an oxygen atom to connect to the metal—a chemical bond that is more reversible because it’s weaker. That change should encourage growth in the horizontal plane.
Based on theoretical analyses and calculations, the team identified a ligand molecule with those characteristics: hexahydroxy-tetraazanaphthotetraphene, or HHTT. Initial experiments with the HHTT ligand were encouraging: The researchers were able to grow larger single crystals than they could using their previous ligand.
But they could add still one more factor to their MOF design toolkit: They could use different metals with their HHTT ligand. The metal can affect the connections between the stacked layers, thereby changing the rate of vertical growth.
To explore the effect of using different metals, they tried combining the HHTT ligand with cobalt, nickel, copper, and magnesium. The result was MOF materials that were related to one another but had slight structural differences. “Using our diffraction techniques, we could actually tell the very minute differences between all of these materials,” says Dincă.
Moreover, they could stabilize the growth to produce and isolate specific single-crystal structures on demand. The images below are scanning electron microscopy (SEM) micrographs of single crystals grown using the HHTT ligand. By optimizing the synthesis conditions, the researchers were able to make high-quality, relatively large single crystals of copper-HHTT in the form of plates (left) and of cobalt-HHTT in the form of rods (right).

Single MOF crystals grown on demand with selected shapes Using a specially selected organic ligand combined with copper or cobalt metal ions, collaborators at MIT and other institutions grew the high-quality single crystals shown in these scanning electron microscope images. By optimizing the synthesis conditions, the researchers could isolate the copper-based crystals as plates (left) and the cobalt-based crystals as rods (right). The hexagonal shape of the rods—visible in the end views in the inset—reflects the hexagonal shape of the individual metal-ligand building blocks. Images courtesy of the researchers and edited by MIT News
Dincă notes that each of the rods in the right-hand image is made up of thousands of metal-ligand units that are connected side by side to form hexagonal sheets, which then stack up to form a hexagonal rod, as is evident in the end-on views in the inset image. “That’s called crystal morphology,” explains Dincă. “The crystal shape often takes on what the molecular shape is.” Each rod is about 20 microns in diameter and 200 microns in length—large enough to be characterized using single-crystal X-ray diffraction analysis.
The SEM micrograph provides physical images of the rod-shaped crystals. So their size and hexagonal shape together confirm that the new metal-ligand combination yields both greater horizontal growth and a larger overall crystal size.
Stacking, slippage, and electrical conductivity
The ability to grow large crystals and examine them using a variety of diffraction techniques creates another opportunity: The researchers could now examine a morphology of interest for controlling electrical conductivity, namely, “slippage” between adjacent layers of their MOF sheets.
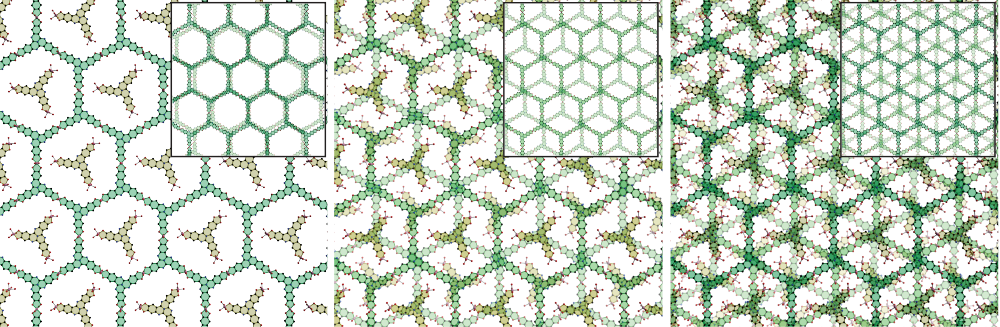
Stacking arrangement of MOF sheets based on analysis of cobalt-HHTT These illustrations represent the view looking vertically down through the stacked layers that make up this three-dimensional structure. The left-hand image looks only at the top layer. If all the sheets below it were perfectly aligned, they would be eclipsed by this top layer and not be visible. But in this structure, the underneath layers are slid or twisted relative to the top one. As a result, in the view through two or three layers—shown in the middle and right-hand image, respectively—the open pores visible in the top layer are eliminated. The researchers’ experimental measurements show that stacking arrangement affects electrical conductivity. Reprinted by permission from Springer Nature ©2020
The figures above demonstrate the concept. The illustrations are representations based on X-ray diffraction analysis of a cobalt-HHTT crystal. The views look vertically down at the three-dimensional structure. The left-hand illustration shows the view when looking at only the top layer. If all the layers were stacked in perfect alignment, with the metal-ligand units centered on top of one another, this top layer would eclipse the layers underneath. The view down through all the stacked layers would look like the top layer, including the open pores. However, in this structure, the underneath layers are slid or twisted relative to the top layer, resulting in so-called staggered packing. The middle and right-hand illustrations show the view through two and three layers, respectively. In both cases, the open pores that occur in the top layer are eliminated.
Three-dimensional materials like MOFs can conduct electricity along their horizontal sheets or perpendicular to those sheets. MOFs generally conduct more easily horizontally than vertically. But slippage may alter that tendency—and may offer another way to fine-tune MOFs for particular applications.
To explore that possibility, Dincă and his team fabricated samples using their HHTT ligand and their four metals, producing two versions of each sample—one with eclipsed packing and the other with staggered packing. They then measured the horizontal and vertical conductivity of the eight samples.
They concluded that whether layers were eclipsed or staggered did affect conductivity. For the most part, conductivity in the horizontal plane was higher than it was in the vertical direction in both versions, as expected. But in general, both conductivities—horizontal and vertical—were higher with eclipsed packing than with staggered packing. So the eclipsed packing encouraged electron flow in both directions.
The researchers highlight some other results of note. With the eclipsed version of copper-HHTT, the observed horizontal conductivity compared favorably with the highest conductivity reported in any MOF. And with nickel-HHTT, the eclipsed version was 10 times more conductive than the staggered version. Vertical conductivity was also correlated with closer stacking of the layers. With almost all of their samples, vertical conductivity became higher as the stacked layers got closer together.
“Clearly, determining the kind of slippage you have is very important for understanding overall charge transport,” says Dincă. “How positive and negative charges travel along a single plane and jump from one plane to another is very much determined by what the slippage is and by the interlayer distance.”
Putting MOFs to work
Dincă and his international team have now established a clear correlation between the structural details and the properties of conductive MOFs. Their methods and results provide other researchers and device designers with guidelines for developing MOFs tailored for specific uses.
But inside Dincă’s lab at MIT, the fundamental work continues, with students and postdocs finding ways to grow MOFs for a variety of practical devices. For example, they have demonstrated a new class of microporous electrodes for use in electrical energy storage devices such as batteries and supercapacitors. They are devising new methods for synthesizing and depositing MOF thin films on substrates for possible use as electrocatalysts and for making MOF-based membranes, some specially designed for gas separation—now an energy-intensive process in many industries. They are also designing MOFs with electronic properties that should prove useful in solar energy conversion and light harvesting. And they are making fluorescent MOFs that could detect certain molecules and signal their presence by emitting fluorescent light.
Dincă believes that MOFs are uniquely suited for addressing important societal challenges in transportation, pollution control, energy conversion and storage, and other areas where traditional materials are reaching the limits of their performance. “With MOFs, the possibilities in terms of structure and function are virtually endless,” says Dincă. He and his team hope to explore as many of those possibilities as they can.
This work was led by MIT Professor Mircea Dincă and involved collaborators from MIT’s departments of Chemistry, Biology, and Electrical Engineering and Computer Science; Peking University and the Shanghai Advanced Research University in China; Stockholm University in Sweden; the University of Oregon; and Purdue University. This research was supported by the U.S. Army Research Office and by Automobili Lamborghini. More information can be found in:
J.H. Dou, M.Q. Arguilla, Y. Luo, et al. “Atomically precise single-crystal structures of electrically conducting 2D metal–organic frameworks.” Nature Materials, vol. 20, pp. 222–228, November 2020. Online: doi.org/10.1038/s41563-020-00847-7.





