CD45 functions as a signaling gatekeeper in T cells
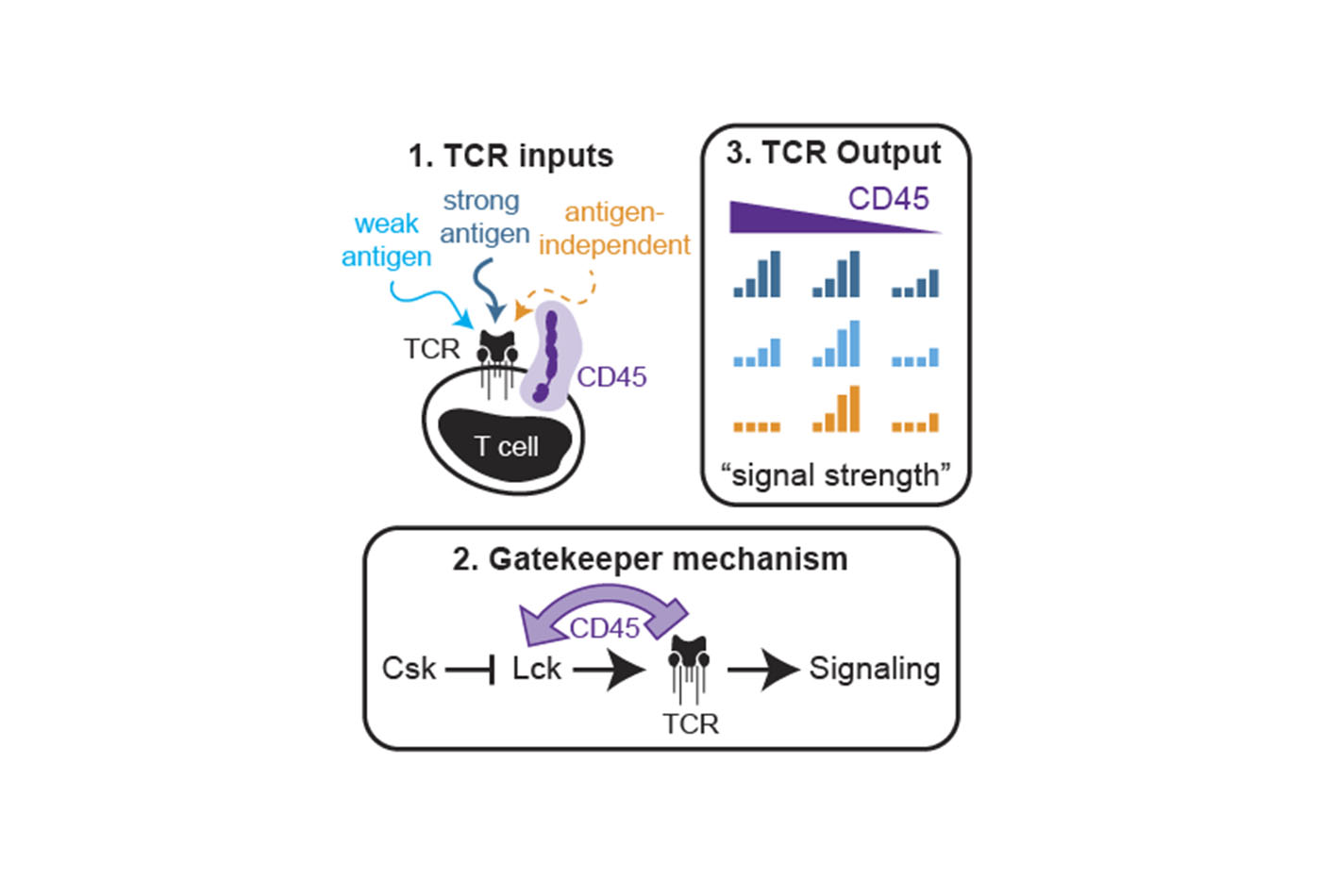
Research from the Chakraborty Group reveals CD45 activates Lck by opposing the negative regulatory kinase Csk.
Professor Arup Chakraborty, along with Adam Courtney, Alexey Shvets, Wen Lu, Gloria Griffante, Marianne Mollenauer, Veronika Horkova, Wan-Lin Lo, Steven Yu, Ondrej Stepanek and Arthur Weiss recently published a research paper in Science Signaling on CD45’s function as a signaling gatekeeper in T cells.
T cells require the protein tyrosine phosphatase CD45 to detect and respond to antigen because it activates the Src family kinase Lck, which phosphorylates the T cell receptor (TCR) complex. CD45 activates Lck by opposing the negative regulatory kinase Csk. Paradoxically, CD45 has also been implicated in suppressing TCR signaling by dephosphorylating the same signaling motifs within the TCR complex upon which Lck acts. The researchers sought to reconcile these observations using chemical and genetic perturbations of the Csk/CD45 regulatory axis incorporated with computational analyses. Specifically, they titrated the activities of Csk and CD45 and assessed their influence on Lck activation, TCR-associated ζ-chain phosphorylation, and more downstream signaling events. Acute inhibition of Csk revealed that CD45 suppressed ζ-chain phosphorylation and was necessary for a regulatable pool of active Lck, thereby interconnecting the activating and suppressive roles of CD45 that tune antigen discrimination. CD45 suppressed signaling events that were antigen independent or induced by low-affinity antigen but not those initiated by high-affinity antigen. Together, their findings reveal that CD45 acts as a signaling “gatekeeper,” enabling graded signaling outputs while filtering weak or spurious signaling events.