MIT Chemists Capture Elusive Boron-Oxygen Ring
This discovery could expand the chemistry of boron-based reagents, providing new tools for oxidation reactions in synthesis and materials science.
Oxygen is a cornerstone of chemistry, largely because it is so good at building the organic molecules that make up our world. Some oxygen-based compounds, called peroxides, are famous for being highly reactive—they act like “oxygen delivery trucks,” transferring atoms to other molecules. This process is essential for everything from creating new medicines to industrial manufacturing.
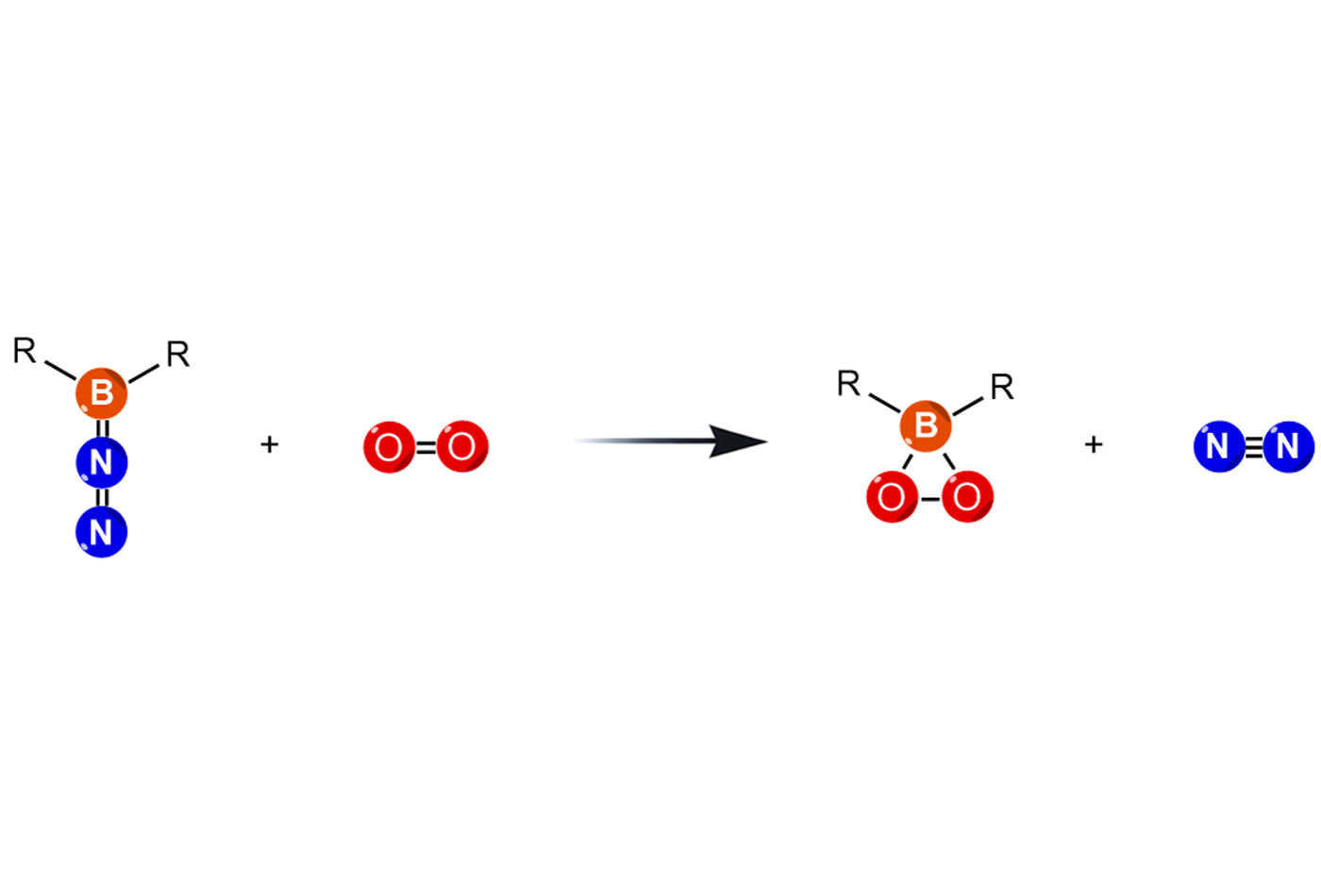
In a study recently published in Nature Chemistry, researchers from the labs of Professor Christopher C. Cummins and Professor Robert J. Gilliard, Jr. have revealed a brand-new type of peroxide containing boron. This molecule, called a dioxaborirane, represents a breakthrough in a field where such structures were long-proposed but considered too unstable to actually isolate.
The “Room Temperature” Breakthrough
The dioxaborirane forms when a specially engineered boron molecule reacts with oxygen gas. What makes this discovery remarkable is that the reaction happens almost instantly at room temperature. Usually, creating strained oxygen-containing rings like this requires extreme, “punishing” conditions—like freezing temperatures or high pressure—to keep the molecule from falling apart.
Using advanced tools like crystallography and computational modeling, the team proved the existence of a highly strained, three-membered ring made of one boron and two oxygen atoms.
A Molecule with Two Personalities
The most exciting part of the discovery is how the molecule behaves. Depending on its electrical charge, it acts in two very different ways:
- The Builder: It can donate oxygen atoms to help construct new chemical compounds.
- The Trapper: It can react with carbon dioxide, potentially offering a new way to capture and transform greenhouse gases.
“By showing that these compounds can be generated under mild conditions, our work opens the door to entirely new types of chemistry,” said Chonghe Zhang, the first author of the paper and a graduate student co-advised by Cummins and Gilliard. “In the long term, these findings could provide us with powerful new tools for oxidation reactions in synthesis and materials science.”